What is Electromagnetic Radiation?
Produced by the nuclear cauldrons of stars and all matter in the cosmos, energy in the form of electromagnetic radiation permeates our entire universe. Every second of every day we are bombarded with and surrounded by electromagnetic radiation; some bounces off of our bodies, some passes through us, and some we absorb, but most goes undetected and unperceived.
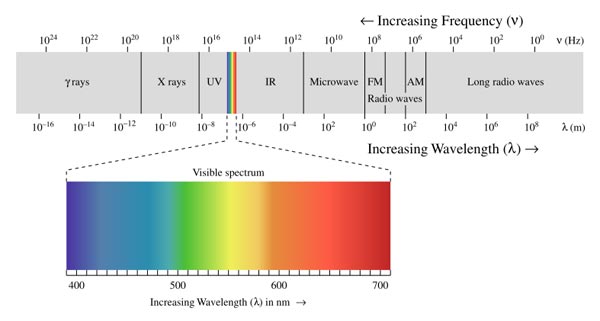
Electromagnetic energy travels at the speed of light (2.9x10^8 m/s or 180,000 miles/sec) in the form of a wave. In fact, we classify electromagnetic energy according to its wavelength. Wavelength is defined as the distance between two corresponding points on two consecutive waves. Electromagnetic wavelengths range in scale from that of an atomic nucleus (gamma rays) to that of a small planet (radio waves).
What is the visible light spectrum?
A tiny fraction of electromagnetic radiation is visible to the human eye and perceptible to the brain. Only the portion of the electromagnetic spectrum that makes it through our corneas and is absorbed by our retinas is perceived as color and light. Often expressed in nanometers (nm) or one billionth of a meter, the wavelengths of the visible spectrum lie between 400 and 700nm, with red light at the longer end of the spectrum and violet light at the shorter end.
The colors in the light visible spectrum
A common acronym used to remember the order of colors in the visible spectrum is "ROY G BIV" (red, orange, yellow, green, blue, indigo, and violet). Just below the visible spectrum from 1 to 400nm lies ultraviolet (UV), while just above from 750nm to 1mm lies infrared (IR).
What role does light play in human vision?
Light plays a crucial role in human eyesight, as it is the fundamental element that, through interacting with the various layers and structures of the eye, enables us to see. Here's how it works:
1, Light Enters the Eye: Vision begins when light rays are reflected off an object and enter the eyes through the cornea, the transparent front layer of the eye. The cornea bends or refracts the rays that pass through a watery fluid, further focusing them.
2. Lens Focusing: After passing through the cornea, light travels through the pupil, the black part in the center of the eye. The iris, which is the colored part of the eye, controls the size of the pupil and thus the amount of light that can enter. Behind the pupil is the lens, a clear part of the eye that further focuses light, fine-tuning it to ensure it hits the right spot on the retina.
3. Image Formation on the Retina: The light rays then reach the retina, a layer of light-sensitive cells (rods and cones) at the back of the eye. The retina converts light rays into electrical impulses. Rods are responsible for vision at low light levels (scotopic vision) and do not mediate color vision, while cones are active at higher light levels (photopic vision) and are capable of color vision.
4. Transmission to the Brain: These electrical impulses are sent through the optic nerve, a bundle of more than a million nerve fibers, to the brain. The brain interprets these signals and creates the images we see.
How Refractive Errors Affect Light in the Eye
- In a normal eye, the cornea and lens have the correct shape and flexibility to refract (bend) light rays precisely onto the retina.
- In eyes with refractive errors, the light is not focused correctly, leading to blurred vision.
- Glasses, contact lenses, or refractive surgery are used to alter the way light rays enter the eye to compensate for the refractive error and focus light directly on the retina.
How Corrective Lenses Affect Light in the Eye
Corrective lenses work by precisely altering the path and focus of light entering the eye to compensate for the specific refractive error, thereby allowing the retina to receive a clear, focused image.
Optical wholesale labs work together with opticians and optometrists to produce lenses with the correction that enables people with refractive errors to enjoy improved vision for both near and distant objects.

